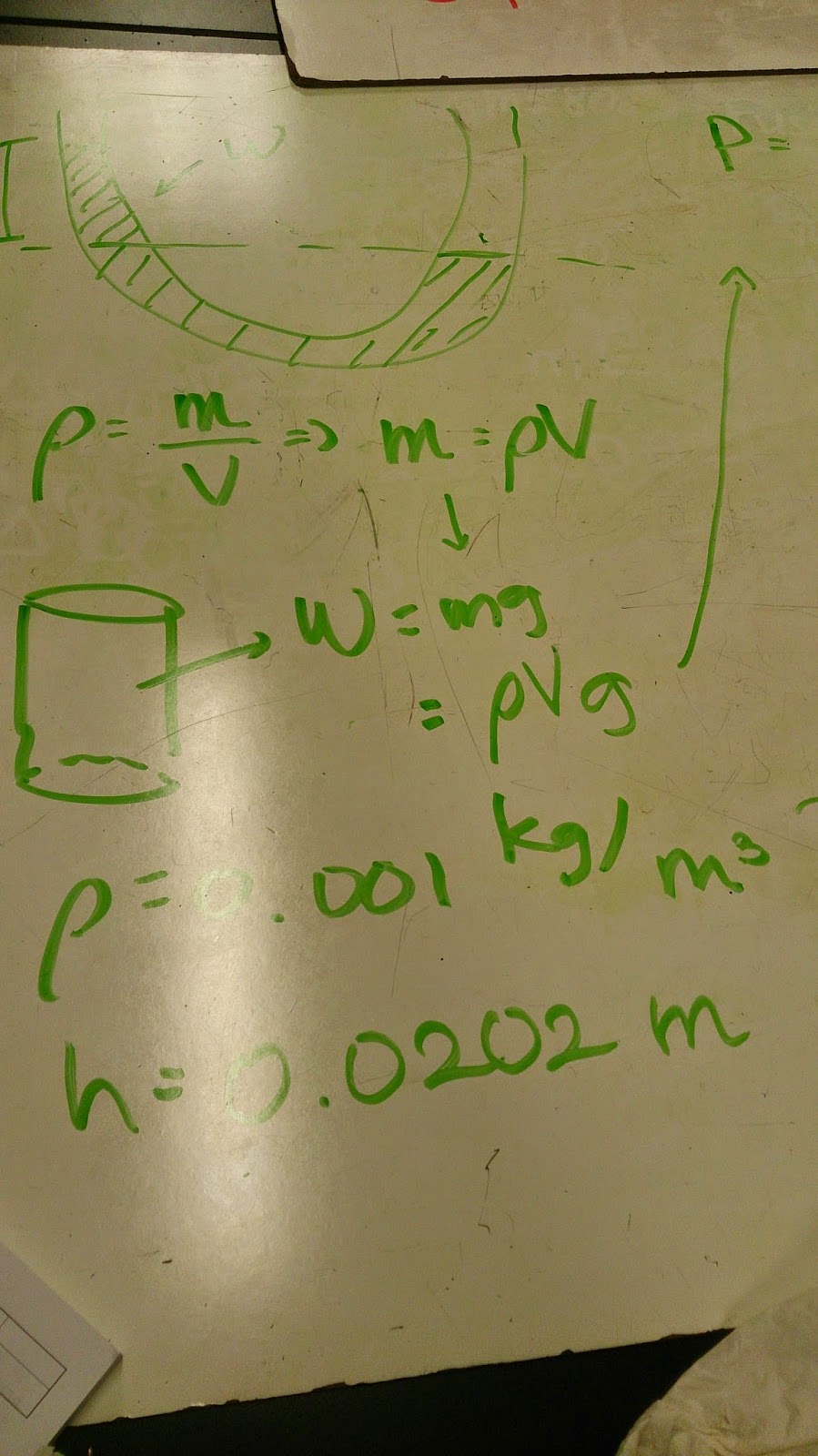Professor Mason
March, 5th
Pressure in Syringe
 First of all, in the class, we did an experiment which includes a friction-less syringe, an empty beaker and a beaker filled with hot water. What we did was to measure the syringe's movement. With the room temperature surrounding the empty beaker, the top part of the syringe slides down slowly. On the other hand, when the empty beaker were surrounded by the hot water inside the other beaker, the top part of the syringe slides up slowly as the pressure makes the atom moves aggressively toward the edges of the empty beaker.
First of all, in the class, we did an experiment which includes a friction-less syringe, an empty beaker and a beaker filled with hot water. What we did was to measure the syringe's movement. With the room temperature surrounding the empty beaker, the top part of the syringe slides down slowly. On the other hand, when the empty beaker were surrounded by the hot water inside the other beaker, the top part of the syringe slides up slowly as the pressure makes the atom moves aggressively toward the edges of the empty beaker.
Work, Heat, and Energy

 The next things we learned in class was about work, heat, and energy. We use the symbol U for internal energy. During a change of state of the system, the internal energy may change from an initial value to a final value We denote the change in internal energy as deltaU = U2 - U1.
The next things we learned in class was about work, heat, and energy. We use the symbol U for internal energy. During a change of state of the system, the internal energy may change from an initial value to a final value We denote the change in internal energy as deltaU = U2 - U1.In this case, work is equal to [Integral of P*dV] or [P*Vo*Beta*delta T]. P is also equal to [2/3 (N*K)/V]

Root Mean Square Velocity
It is represented by the equation: Vrms =[squareroot of ((3*R*T)/m)] or [squareroot of ((3*Kb*T)/m)] , where Vrms is the root-mean-square of the velocity, m is the molar mass of the gas in kilograms per mole, R is the molar gas constant, Kb is Boltzmann constant, and T is the temperature in Kelvin. We found out that V total is equal to 3Vx by doing the magnitude of 3 Vs as showed from the picture above.
Fire Syringe
 In class, we also did another experiment, which we used the tool called
fire syringe. By pushing the plunger as quick as we can, the goal to avoid
change in pressure as seen in the video below would succeed because we wanted
to change volume without changing the pressure. The temperature increased
rapidly when we did this. First before we started pushing the plunger, we needed
to collect the data, such as the height of the syringe measured from the bottom
where we placed the cotton at to the part where the plunger ends. Then, we make
some calculations to predict the final temperature. The ignition point of
cotton is about 210*C or 483K. After we got all the data, we started pushing
the plunger as fast as we can in order to light up the cotton. Based on the
calculation picture below, we found out the final temperature to be 2860.8K.
Thus, the cotton did light up as shown in the video as well.
In class, we also did another experiment, which we used the tool called
fire syringe. By pushing the plunger as quick as we can, the goal to avoid
change in pressure as seen in the video below would succeed because we wanted
to change volume without changing the pressure. The temperature increased
rapidly when we did this. First before we started pushing the plunger, we needed
to collect the data, such as the height of the syringe measured from the bottom
where we placed the cotton at to the part where the plunger ends. Then, we make
some calculations to predict the final temperature. The ignition point of
cotton is about 210*C or 483K. After we got all the data, we started pushing
the plunger as fast as we can in order to light up the cotton. Based on the
calculation picture below, we found out the final temperature to be 2860.8K.
Thus, the cotton did light up as shown in the video as well.